Aggregation
Protein immunogenicity remains an important concern for biotherapeutics. Among the several factors playing a role in immunogenicity, the presence of aggregates is considered an important product-related factor that may increase the risk of an immune response. Although little is known about which aggregate species trigger the immune system, it is believed that aggregates are more easily recognized by the immune system than the native parent protein.
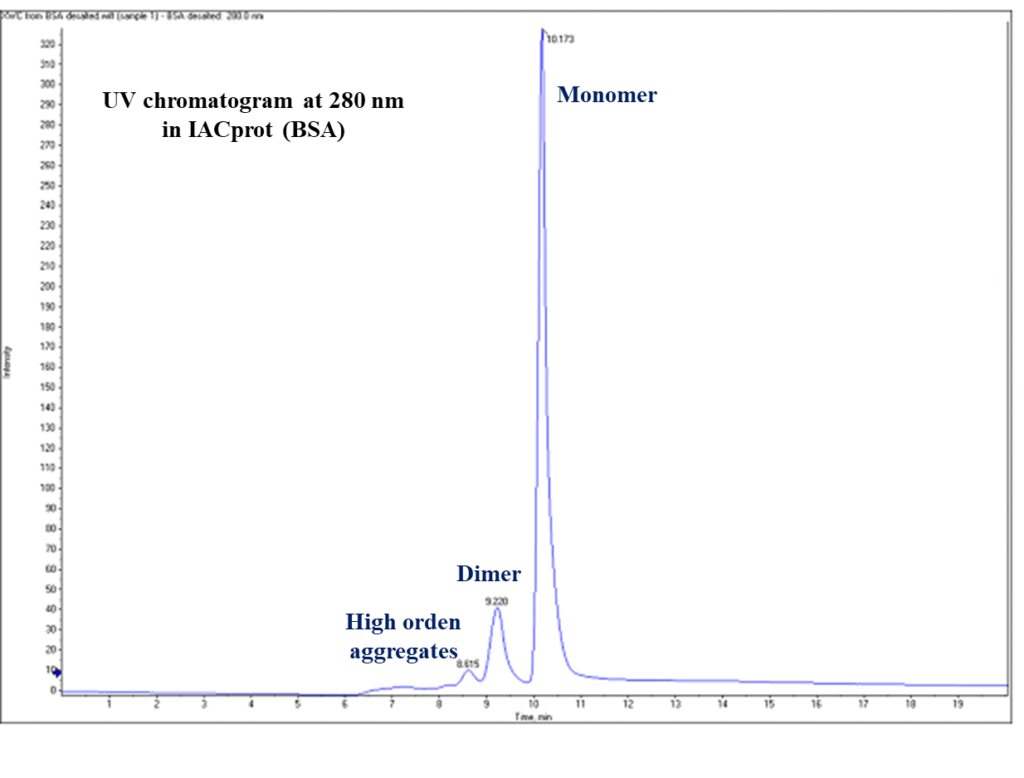
The aggregates are usually dimers, trimers, and sometimes higher-order aggregates. The aggregation profile must be monitored, both during the production process and during the storage period as required in ICH Q6B guidelines. AMSbiopharma scientists have designed a strategy for aggregation protein/monoclonal antibody determination based on HPLC-DAD-TOFMS analysis separating by size exclusion chromatography (SEC) the protein monomer, dimer and the high order aggregates.
Moreover, with the novel online high-resolution mass spectrometry detection the molecular weight of each species can be determined confirming if the analyzed fragment are monomer, dimer or other aggregates. Using the peak areas in the 280 nm UV chromatogram, the relative concentration of monomers, dimers and other aggregates of your biopharmaceutical can be determined.