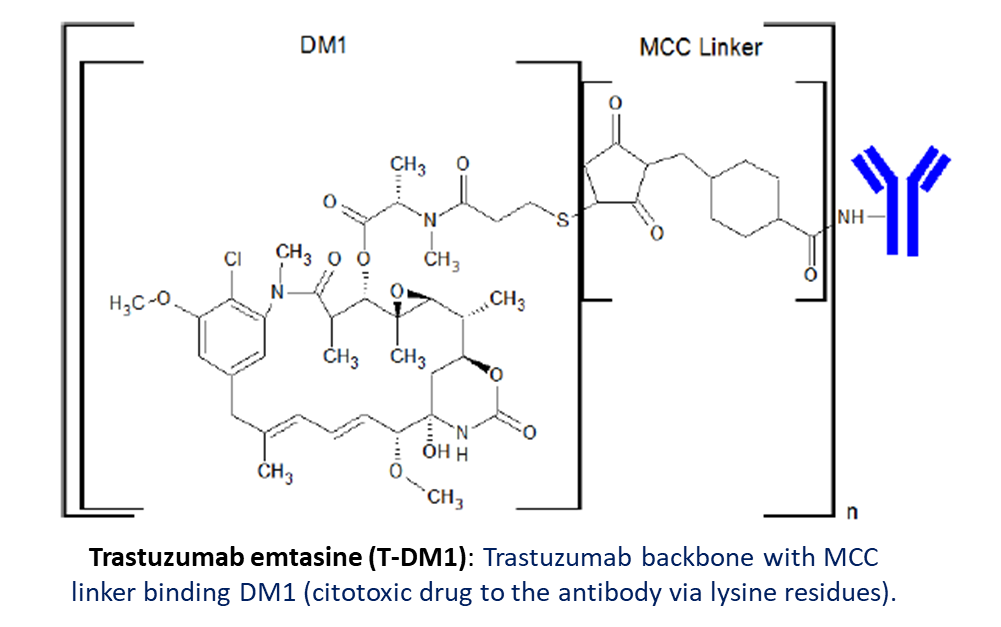
Drug to antibody ratio (DAR)
Drug antibody ratio (DAR) is an important parameter of ADC and refers to the average drug to antibody ratio for a given preparation of antibody drug conjugate (ADC). The significance of DAR in biotherapeutics is a primarily as a measure of drug loading for an ADC.
For a heterogeneous population of drug loaded antibody, the distribution of drug can impact the therapeutic efficacy of an ADC and properties such as drug clearance, pharmacokinetics, and biodistribution. It is common to see DARs of up to 8 drugs per antibody when linkage is through Cysteine or Lysine residues.
So, due to this impact, AMSbiopharma have developed a specific analytical service to determine DAR with high precision for your ADC biotherapeutic. For drug to antibody ratio (DAR) calculation, the ADC is analysed in the intact form by electrospray ionization and high resolution mass spectrometry (ESI-TOFMS) coupled with reverse phase HPLC. The obtained multiple charged mass spectra enable the determination of the different DAR species present in your ADC biopharmaceutical.
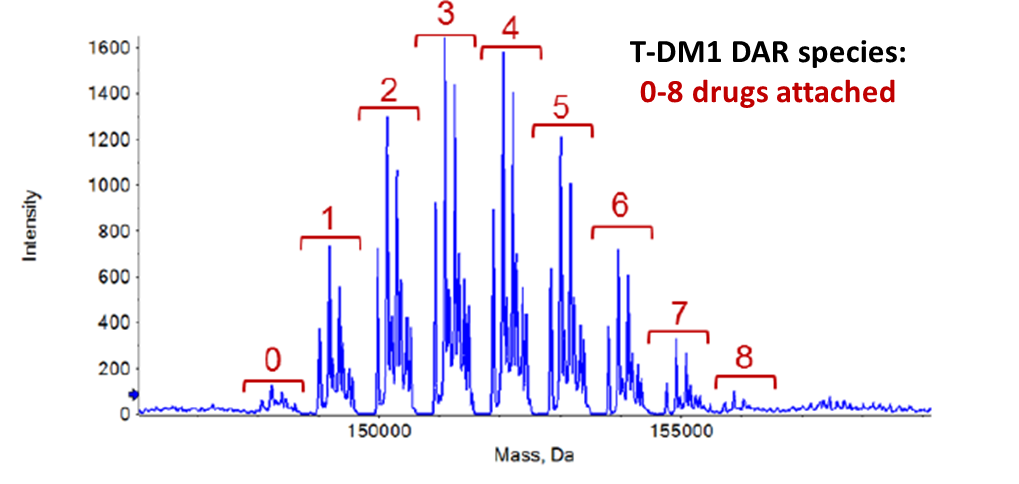
Finally, DAR is calculated employed a specific software and based on the reconstructed peak area distribution of the different DAR species.
Moreover, if you need to know the specific binding site of your biopharmaceutical, AMSbiopharma offers you an additional service based on a peptide mapping analysis: A tryptic digestion of your ADC, followed by analysis of the resulting peptides by HPLC-QTOFMS. The high resolution mass spectra and fragmentation spectra allow to know in which lysine or cysteine residues the drug is linked.

