Clinical efficacy of Antibody Drug Conjugates (ADC) is determined by fine-tuning combination of tumor antigen, targeting antibody, cytotoxic payload and conjugation strategy. ADC binds to tumor target cell surface antigens triggering a specific receptor mediated internalization. The internalized ADCs are decomposed to release cytotoxic payloads inside the tumor cell.
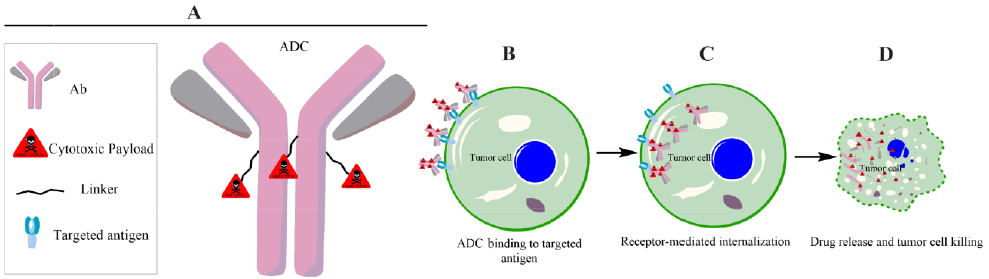
AMSbiopharma (from AMS Labs) provides a particular key characterization analysis for ADCs such as: Drug antibody ratio (DAR) and Drug conjugation size.
Moreover, as stated in the ICH guideline Q6B, AMSbiopharma provides a comprehensive catalog of services required for the characterization of a biopharmaceutical.
| Cookie | Description | ||
|---|---|---|---|
| Cookie | Description |
|---|---|
| Cookie1 | Cookie1 Prueba |
| cookielawinfo-checkbox-necessary | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-non-necessary | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Non Necessary". |
| test_cookie | Test Description |
| viewed_cookie_policy | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |