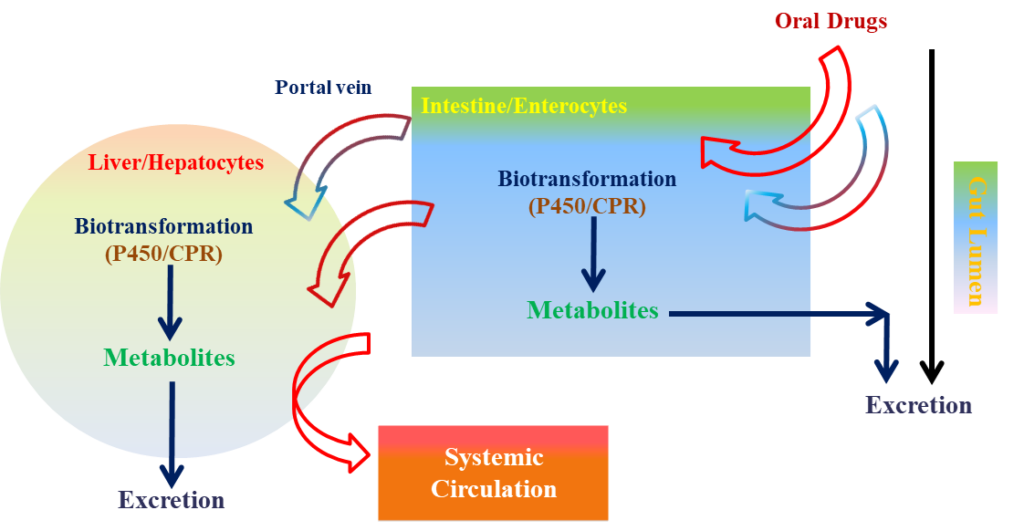
Drug-drug interactions (DDIs) are one of the commonest causes of medication error in developed countries, particularly in the elderly due to poly-therapy, with a prevalence of 20-40%. In particular, poly-therapy increases the complexity of therapeutic management and thereby the risk of clinically important DDIs, which can both induce the development of adverse drug reactions or reduce the clinical efficacy. For example, if a certain component of drug A changes the activity of enzymes or transporters involved in the metabolism of drug B, it may either reduce the therapeutic efficacy or enhance the toxicity of drug B, leading to severe clinical consequences.
Cytochrome P-450 (CYP-450) is primarily responsible for the oxidative metabolism of drugs in humans. It is not a single enzyme, but is actually a family of hemoproteins present in numerous species.
Drugs that exert an inhibition or an induction on CYP450 need to be detected and eliminated quickly in the early stages of its development. Furthermore, it is important to detect whether a compound is a CYP substrate and to be able to associate the specific isoform involved with it to predict undesirable DDIs.
AMSbiopharma has established a cytochrome P450 service that includes: CYP inhibition assay and CYP induction assay.
- CYP inhibition assay: The seven main cytochrome P450 isoforms (CYP1A, CYP2B6, CYP2C9, CYP2C8, CYP2C19, CYYP22D6 and CYP3A4) are investigated in AMSbiopharma P450 inhibition assay. In this assay, isoform-specific substrates are incubated individually with liver microsomes and a range of test compound concentrations. At the end of the incubation, the decrease in the formation of metabolites is compared to vehicle control (positive control inhibitor).
- CYP induction assay: AMSbiopharma CYP induction assay identifies the potential of test compounds to induce CYP1A2, CYP2B6 or CYP3A4 in cultured human hepatocytes by evaluating catalytic activity. If a cytochrome P450 enzyme is induced by a test compound it may both increase the metabolism of itself (autoinduction) or a concurrent therapy, and so reduce levels resulting in a decrease in efficacy. Three probe substances for catalytic activity are used: Phenacetin (CYP1A2), Bupropion (CYP2B6) and Midazolam (CYP3A4) and the quantification of acetaminophen (CYP1A2), hydroxybupropion (CYP2B6) and 1-hydroxymidazolam (CYP3A4).